Radiotherapy
How it works
Most radiotherapy is given using a beam of high-energy X-rays which are more powerful than those used for taking ordinary X-ray pictures and given in longer bursts – for example, a minute or so, compared with less than a second for diagnostic pictures. These beams shed their energy into the cells of the tissues through which they pass. You have to lie still during treatment, but you don’t feel anything and from your point of view the only difference from having an ordinary X-ray is that radiotherapy lasts longer. You don’t become radioactive.
The target
Radiotherapy targets the DNA in the cell nucleus because, if this is damaged sufficiently, cells will lose the ability to replicate. If cells in a cancer that die naturally are not being replaced by new ones, not only will the cancer stop growing, it will also become smaller and will eventually wither away and disappear.
Normal cells are affected to some extent by radiotherapy too. Fortunately, normal cells in general have a greater ability to repair radiation damage than cancer cells. The therapy is done in such a way as to ensure that cancer cells receive the highest dose of radiation, while minimising as far as possible the amount that reaches nearby normal cells. This may be done by using crossed beams focused on the tumour or by shielding some of the normal tissue (see below).
How cancers respond to treatment
Cancers vary considerably in their sensitivity to radiotherapy: some types are more likely to be totally eradicated than others. How quickly they respond to treatment is also very variable. Some cancers will continue to shrink slowly and eventually disappear long after a course of radiotherapy has been completed, possibly over many weeks. Others will respond much more quickly, particularly those in which the cells were dividing rapidly before treatment.
In general, smaller cancers have a greater chance of being completely destroyed by radio-therapy than larger ones. This is partly because of the sheer volume of tissue involved, but also because larger tumours tend to outgrow the blood vessels supplying them, resulting in a reduced blood supply and thus less oxygen at their centre. Oxygen is very important to the process of DNA damage caused by radiation. Oxygen is carried to the tissues by the red blood cells, so if the tumour has a poor blood supply, or if there is a lowered concentration of red cells in the blood (anaemia), radiotherapy may be less effective. Thus curing anaemia by a blood transfusion can, in certain circumstances, make radiotherapy more successful.
Another reason why radiotherapy may sometimes not achieve complete tumour destruction is that the tumour cells continue to replicate rapidly in between daily treatments. One way of trying to deal with this that is occasionally used in certain situations is to give radiotherapy two or three times per day in a very short and intensive course of treatment. This is known as ‘hyperfractionated’ and ‘accelerated’ treatment.
Another way of enhancing the effectiveness of radiotherapy is to give chemotherapy as well, at the same time. This has, for example, improved cure rates for patients with certain lung, cervical and oesophageal cancers, although the side effects from such combined treatment tend to be more marked.
Radiotherapy can destroy a cancer only if it is possible to target the beams so that they encompass the whole of the tumour. It can be used to treat rather larger portions of tissue than can often be dealt with by surgery, but is nevertheless a fairly localised form of treatment. Another crucial factor is the ‘dose’ of radiotherapy, measured in units called ‘grays’. Some cancers will respond well to relatively low doses whereas others may need very high doses if they are to be destroyed.
Radiotherapy is usually given in quite high dosage (radical radiotherapy) when it is given by itself with the intent of completely destroying a cancer.
A slightly lower dose is given when radiotherapy is being given as an adjuvant treatment to prevent a cancer returning after surgical removal. The dosage is usually considerably lower when the treatment is palliative. In this situation the aim is to cause sufficient tumour shrinkage to alleviate symptoms, but not to eradicate the cancer completely.
Side effects of radiotherapy
If you are receiving low-dosage radiotherapy you may not notice any side effects whatsoever, but those receiving high-dose treatment can usually expect to experience some. What they are, and their severity, depend on the part and amount of the body being treated, the dose of radiotherapy and also on how sensitive a particular individual is to treatment.
The most common side effects occur during or immediately after treatment and are short lasting, when they are described as being ‘acute’ They usually disappear quite quickly after treatment. A few people may develop so-called ‘late’ side effects several months or even sometimes several years after treatment. These effects can be long lasting (‘chronic’) or even permanent. It is fairly unusual for long-lasting effects to be particularly troublesome, but small risks are often justifiable when aiming for cure.
Short-term effects
The normal tissues that tend to be most sensitive to radiotherapy in the short term are those where the cells are normally dividing quite rapidly to replace those lost by wear and tear. These include the skin and the membranes lining the mouth, throat, oesophagus, bowel and bladder. Inflammation, soreness, diarrhoea and urinary frequency are thus quite common side effects, depending on the part of the body being treated. Some patients get skin soreness rather like sunburn, but unless a superficial tumour is being treated it is now unusual for this to be severe, as modern radiotherapy beams can deliver the maximum dose deep, away from the surface.
Other quite common acute side effects include tiredness, nausea (particularly if the upper abdomen is being irradiated), and hair loss if treatment beams go through the scalp. This is not usually permanent, although there may be patches where the hair does not regrow if radiotherapy is being given in fairly high dosage for a brain tumour. Bone marrow is also very sensitive to radiation but this is usually only a problem in the unusual event of treatment being given to a large part of the body containing a large proportion of the total amount of marrow.
Before having radiotherapy you will be warned about the more common side effects and probably given advice about how, for example, to care for your skin or change your diet to try to prevent the reaction becoming at all severe. If your abdomen or pelvis is being treated you may be recommended to cut out high-fibre foods, including fruit and green vegetables, until the reaction has settled. This is because they can make diarrhoea worse. If necessary, you may be given one of the various medications available to lessen side effects. Occasionally it is necessary to interrupt the course of treatment for a week or so to allow side effects to settle.
Tiredness and other acute side effects of treatment can sometimes interfere considerably with normal living, but many people have only slight if any side effects. You may well be able to continue working during your treatment and your doctor may encourage you to do so if you feel up to it. While it’s sensible not to push yourself too much if you feel tired or unable to carry on with your normal life, there is usually much to be said for remaining fairly active if at all possible, unless advised to the contrary. However this may not be appropriate for everyone and if you are unsure you should discuss this with your oncologist or the radiographers treating you.
Long-term effects
Tissues that tend to be sensitive to radiotherapy in the longer term include the lungs, kidneys, the eye lenses (where cataracts may form), and the testes and ovaries (possibly resulting in infertility). However, significant damage to these tissues can usually be avoided by careful treatment planning or, if attempting to preserve fertility, occasionally by surgically moving (‘transposing’) the ovaries a little, so that they are missed by most of the irradiation. If such measures are not possible, deep frozen storage or ‘cryo-preservation’ of sperms, embryos or eggs may be appropriate for those who may want to have children in the future.
Other occasional long-term effects include arm swelling or lymphoedema as a result of treatment to the armpit after surgery for breast cancer, and bowel damage following radiotherapy for cancer of the cervix. In the past some women having radiotherapy for left-sided breast cancer experienced long-term effects on their heart, but as a result of modern refinements in radiotherapy planning and delivery this risk is now extremely low.
Radiotherapy can cause mouth dryness if the salivary glands are being treated and this can predispose to both tooth decay and gum disease. Patients at risk for this problem should have a specialist dental assessment before treatment, and be given advice about caring for their teeth in the future. Vaginal dryness can also be a consequence of pelvic radiotherapy and lubricants may be necessary for normal sexual function. Growing tissues are also very sensitive to radiotherapy and this has important implications for treatment for children.
Finally, there is a very low risk that the radiation might itself cause another cancer many years later. However, this risk almost always pales into insignificance when compared with the potential benefit from treatment.
How radiotherapy is given
Treatment from the outside
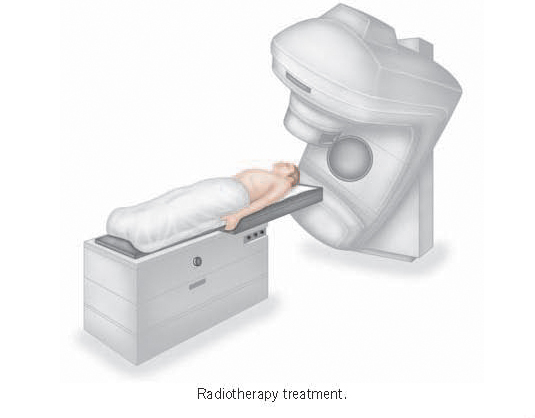
Radiotherapy is usually given to a fairly localised part of the body. It is given mostly using high-energy, deeply penetrating, ‘megavoltage’ X-ray beams produced in large machines called linear accelerators. Sometimes, less penetrating, lower-energy, ‘orthovoltage’ beams produced by much smaller machines are used to treat relatively superficial growths, particularly skin cancers. Electron beams produced in linear accelerators are also used sometimes to treat fairly superficial tissues. All these methods involve beams of radiation being ‘shone’ into the body from outside, and are known sometimes as ‘external’ radiotherapy.
External radiotherapy is given in a ‘treatment room’ which has specially thick walls to prevent radiation escaping from it. The treatment is given under the supervision of the radiographers who control the treatment machine. During the actual treatment the radiographers will stay outside and the patient is alone in the room. This is because if they stayed inside they would themselves receive, over a period of time, a significant cumulative dose from ‘scattered’ radiation. They do keep a close eye on you, however, via closed circuit television or by looking through a special radiation-proof window.
Planning and prescribing treatment
Radiotherapy can vary enormously in complexity. Some treatments are technically quite simple. When treating skin cancers the oncologist merely has to draw on the skin to indicate the area requiring treatment, and then specify the dose, the number of treatments and their frequency. The radiographers can then begin treatment straightaway. The patient has to lie very still in the appropriate position and the radiographers ensure that the beam is directed accurately to the right area and for the correct amount of time to give the required dose.
But most treatments are rather more complex, using at least two and sometimes several radiation beams entering the body from different angles and converging at the site of the cancer or the part of the body to be treated. You may need to have a CT or MRI scan beforehand to define as accurately as possible the position, size and shape of the cancer. Meticulous planning is required. This will involve your lying on a couch in a ‘simulator’, a machine that looks rather like a linear accelerator, but which produces X-ray images or scans of the part of the body to be treated. Physicists using computers then work out how best to arrange the treatment beams so that they will deliver the required dose to the intended target with great accuracy, while keeping the dose to the surrounding normal tissues to a minimum.
Sometimes the beams will be shaped specially to achieve this, using ‘multi-leaf collimators’ inside the treatment machine. Such special shaping of the beams can in certain situations be used to deliver high dose treatment to a volume of tissue that conforms closely and 3-dimensionally to the shape and extent of the tumour. This is known as ‘conformal radiotherapy’. Whatever treatrment technique is chosen, once the oncologist has checked the resulting ‘treatment plan’ and prescribed the treatment, radiotherapy can then begin.
To be certain that the treatment beams are entering the body at the correct positions, ‘reference points’ are often marked on the skin, either with felt-tip pens or as pinprick-sized tattoos. If felt-tip pen marks are used you will be warned not to wash them off! Final accuracy checks may be made on the simulator by taking X-ray images corresponding to the treatment beams, or by producing precise pictures of the tissue being irradiated on the radiotherapy machine itself.
As accuracy is so vital you may be fitted with an ‘immobilisation mould’ or ‘shell’, sometimes also known as a ‘jig’, especially if you are having radiotherapy to the head or neck. This is an individualised transparent plastic mould which fits like a glove over the part of the body being treated and which is fixed to the treatment couch, so stopping you moving even very slightly during treatment. An additional benefit is that the entry points of treatment beams can be marked on the plastic surface without resorting to skin tattoos. Other types of immobilisation devices are also quite often used.
A fairly recent technical development is ‘intensity-modulated radiation therapy’ (IMRT). This is high precision three dimensional conformal radiotherapy in which the beams are not only customized by being meticulously shaped, they are also tailor-made by having variation in the intensity of the radiation across each beam. This further facilitates the delivery of a high dose to the cancer whilst minimising the dose to the surrounding normal tissues. In theory this should result in an increased chance of cure and/or better tolerability. Yet another advance in accuracy can be obtained with ‘image-guided radiation therapy’ (IGRT) in which the radiation beams are adjusted to take account of any slight change in tumour or patient position during a course of treatment and even during individual treatment sessions, for example with breathing. This fairly new development is effectively four-dimensional treatment and it may become increasingly used for cancers in certain parts of the body that are more prone to change in position.
Another highly precise form of treatment is ‘stereotactic’ radiotherapy, also known as ‘radiosurgery’, where very small beams entering from very many different angles are focused in a highly precise manner on a very small part of the body. This is used for some people with small tumours in or near the brain, whose heads are immobilised completely during treatment using a special frame. Radiosurgery is now being used increasingly to destroy small tumours in other parts of the body. It is given with machines such as the Gamma Knife which delivers narrow radiation beams (‘gamma rays’) from a helmet containing a couple of hundred sources of radioactive cobalt, or the CyberKnife – a small linear accelerator which rotates around the patient whilst focusing a narrow beam of high energy X-rays on the tumour.
How long does treatment last?
Although most individual radiotherapy treatments take only a couple of minutes or less to deliver, all the position and other accuracy checks often mean that you are in the treatment room for 10–15 minutes per session.
Sometimes, particularly when the main aim of treatment is to relieve symptoms, only one or two sessions or ‘fractions’ are required. In other instances treatment may continue once a day for perhaps from three up to six or seven weeks. For long courses such as this you will probably have weekends off. However, occasionally a more intensive short programme may be preferred, involving ‘continuous’ hyper-fractionated and accelerated treatment (‘CHART’) two or three times per day including over the weekend.
Treatment from the inside
Some cancers are best treated by placing small amounts of a radioactive ‘isotope’ inside the patient, either within or very close to the growth. This is known as ‘brachytherapy’. The isotope emits rays known as ‘gamma rays’, but which have identical properties to X-rays. This makes it possible to deliver a high radiation dose to the cancer cells, while normal tissues a little further away receive only a considerably reduced dose. This can decrease the chance of troublesome side effects.
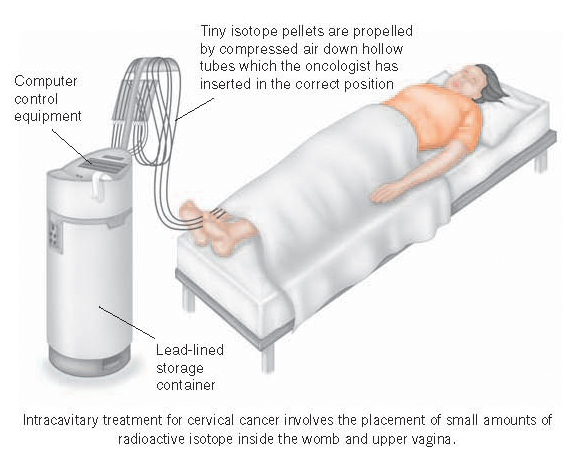
This method is used frequently in the treatment of cancer of the cervix, when the radioactive isotope is inserted into the uterine cavity and upper vagina under anaesthesia. The isotope may be left in place overnight, with the patient in a special room on the ward. However most patients are now treated with a much quicker ‘high-dose rate’ technique using a more powerful isotope, with an individual treatment being completed in a special treatment room in a matter of minutes. This is then repeated a few times over several days.
Such treatments are known as ‘intra-cavitary’, because the source of the radiotherapy is placed within a body cavity. It is quite often given in addition to a course of external treatment. Intracavitary radiotherapy is also being used increasingly as a palliative treatment for patients with lung and oesophageal cancer.
For patients with other growths, for example, some prostate, breast and tongue cancers, small amounts of the isotope may be placed actually within the cancerous tissue, which is ‘implanted’ using special needles or other penetrating devices, under anaesthesia. This is known as ‘interstitial’ brachytherapy and is being increasingly used as an alternative to surgery for patients with small prostate cancers. Intracavitary and interstitial treatments are often given using computer-controlled equipment which propels tiny isotope pellets by compressed air through hollow tubes which have been inserted by the oncologist while the patient is anaesthetised. Once in place in the cavity or inside the tumour, the pellets are left there for the required amount of time. They can then be sucked back into a lead-lined safe in which they are stored safely.
In another type of radiotherapy you may be given an injection of a radioactive isotope or asked to swallow it. It is then carried round the body in the bloodstream. The isotopes used in this form of treatment have a strong tendency to ‘home in’ on certain tissues such as benign or malignant thyroid tissue (radioactive iodine) and bone metastases (radioactive samarium and strontium). These radioactive isotopes emit electrons (also known as ‘beta rays’) which have a very short range, but which can destroy cancer cells when released by the isotope very close to them. As the cell-killing effect is directed predominantly against the cancer cells, these treatments usually cause few side effects.
KEY POINTS
-
Radiotherapy, like surgery, is a localised form of treatment
-
Radiotherapy has the potential to destroy a cancer completely while leaving the surrounding normal cells intact
-
You don’t feel anything during treatment
-
Cancers vary considerably in their sensitivity to radiotherapy
-
High-dose radiotherapy given for cure often carries a risk of long-term side effects, but this is usually very low and well worth taking
-
There have been major technological advances in radiotherapy planning and delivery in recent years, making it easier to give the right dose exactly where it is needed
-
Most patients will benefit from keeping active during treatment